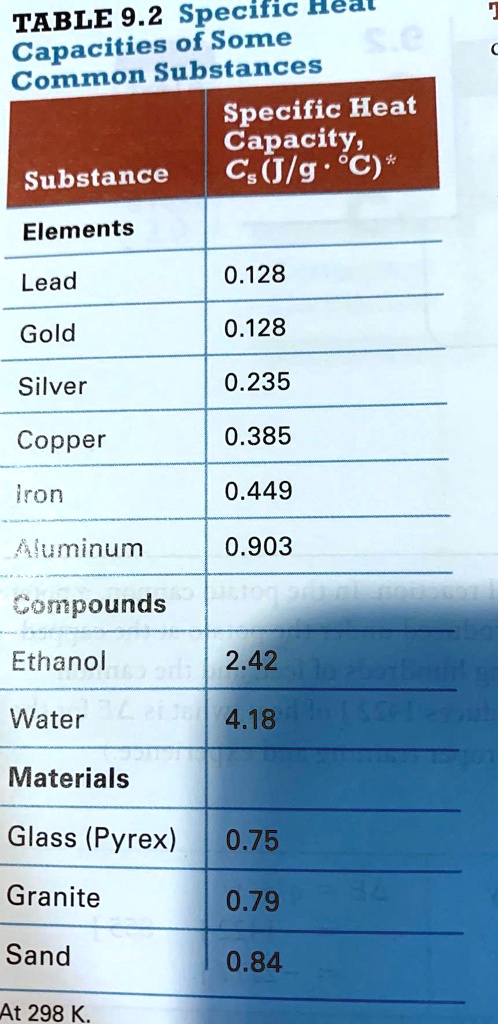
Specific heat capacity of Apiezon N high vacuum grease and of Duran borosilicate glass - ScienceDirect
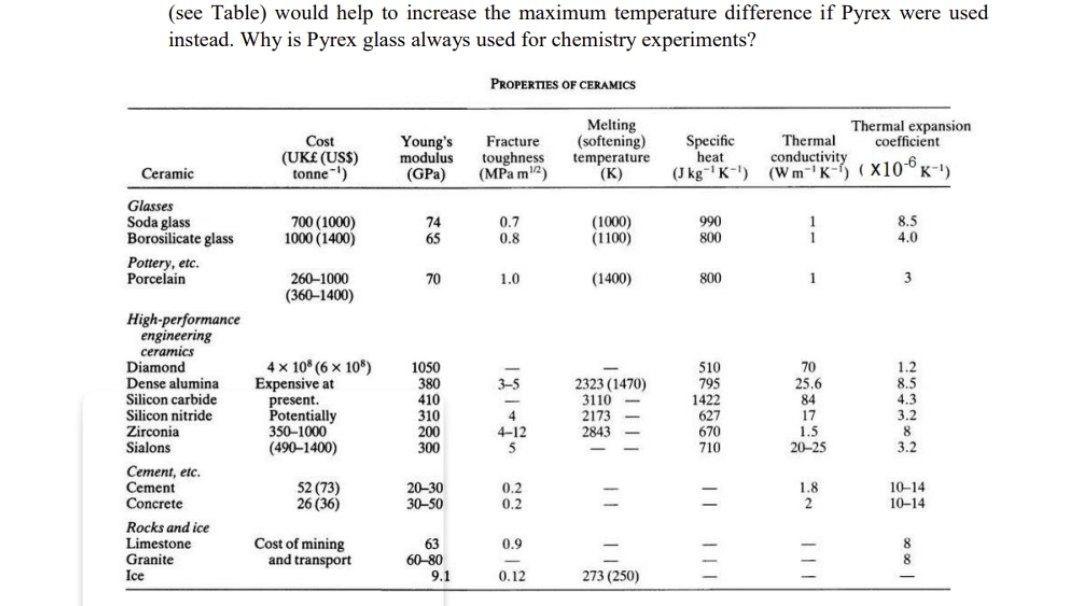
Primary experimental thermal conductivity measurements for Pyrex 7740,... | Download Scientific Diagram

Effect of Composition and Temperature on the Specific Heat of Glass - SHARP - 1951 - Journal of the American Ceramic Society - Wiley Online Library

PHYS 101 Quiz 5.docx - Question 1 2 out of 2 points A bimetallic strip is formed from two materials that have different Selected Answer: | Course Hero
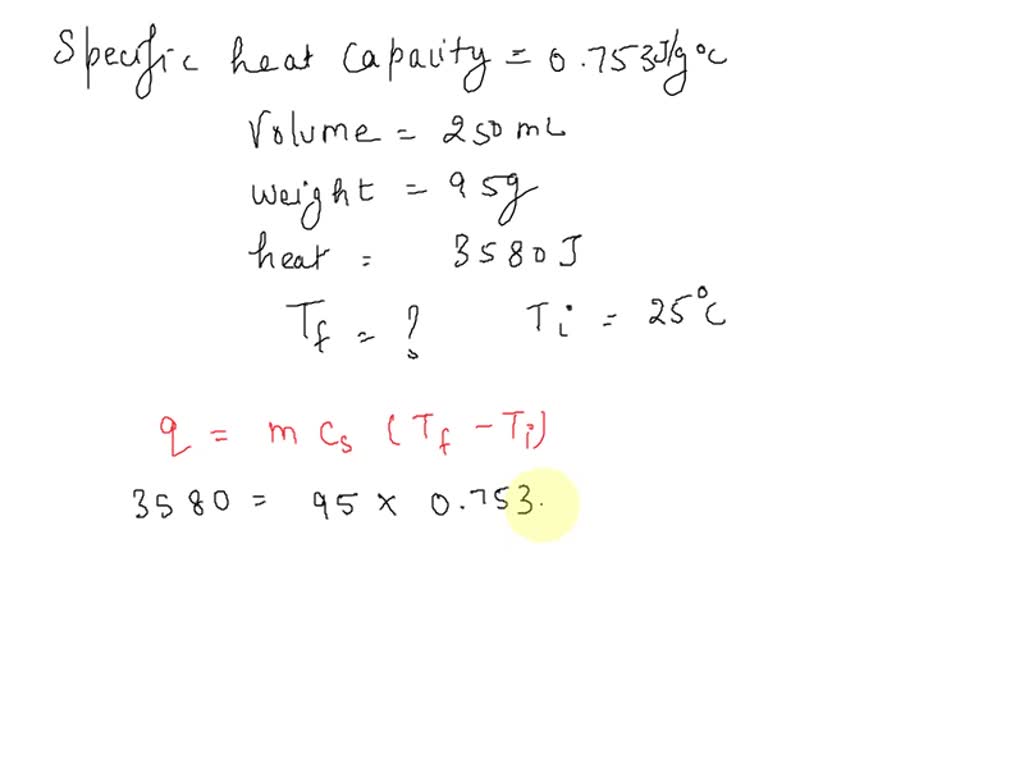
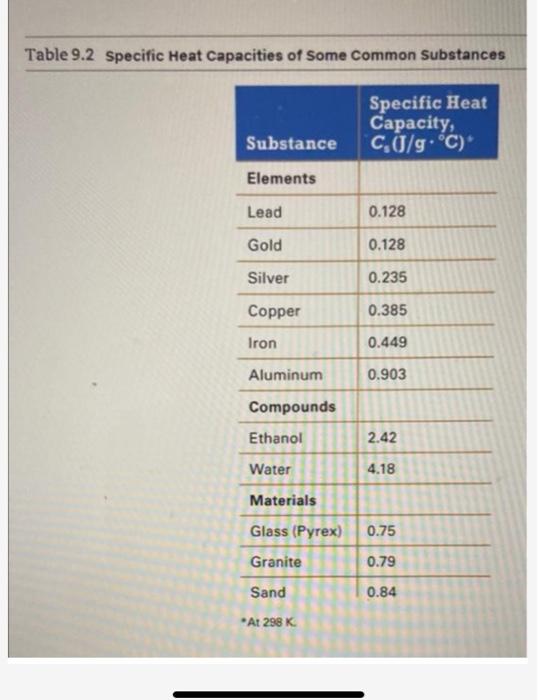
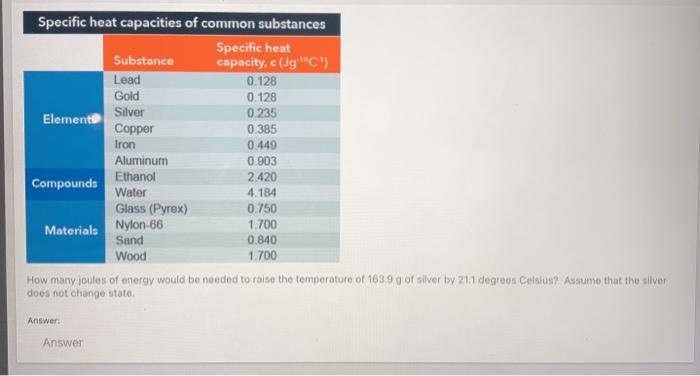
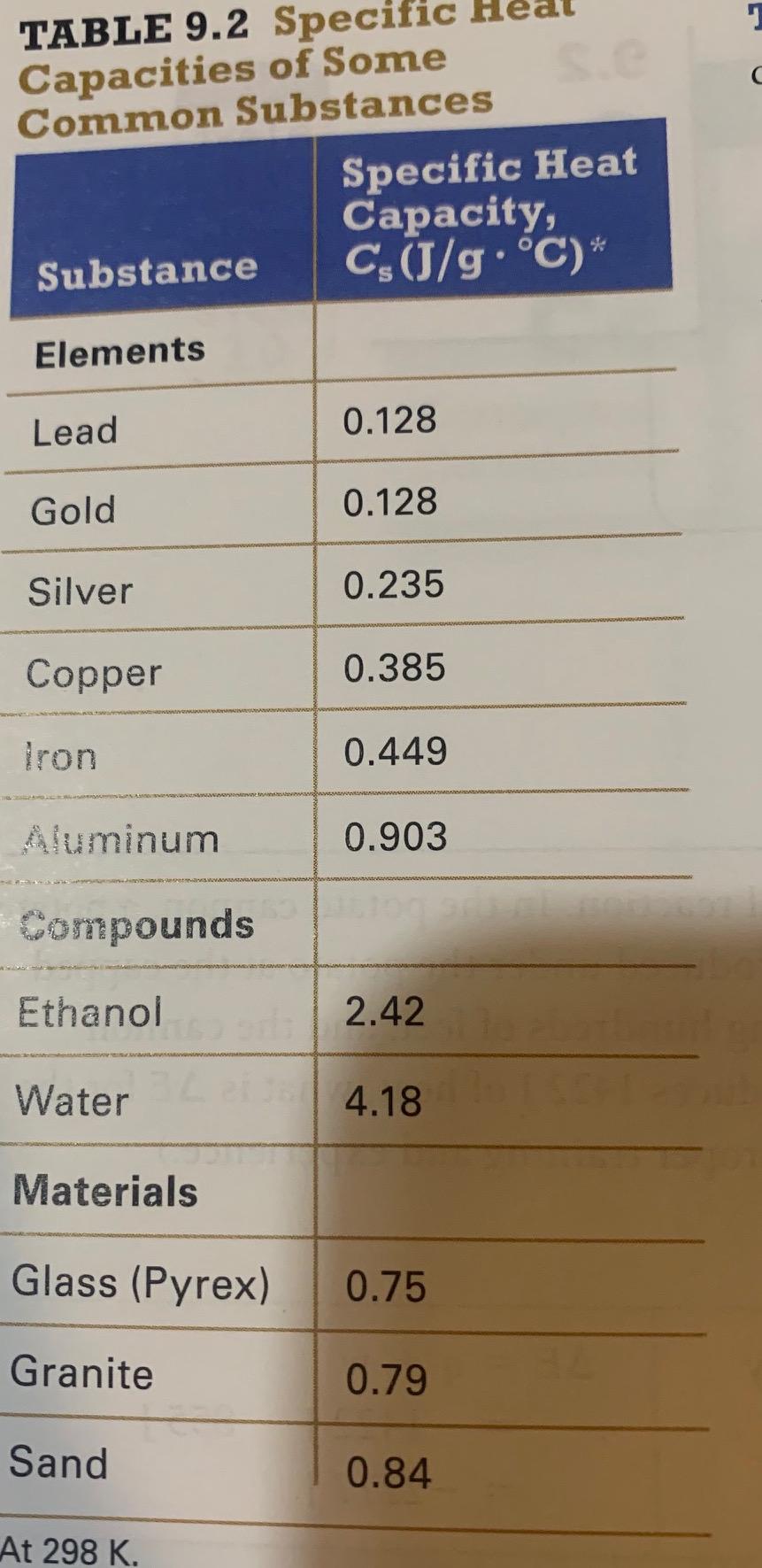
SOLVED: the specific heat of Pyrex glass is 0.753 J/g, °C. A 250 mL beaker weighing 95.000 g was allowed to absorb 3580 J. What is the final temperature of the beaker

Amazon.com: Synergy Trading Pyrex Heat Resistant Glass Container, Measuring Cup, Measuring Cup, Measuring Cup, Jug, Measuring Cup, Oven, Microwave Safe, 16.9 fl oz (500 ml) : Home & Kitchen
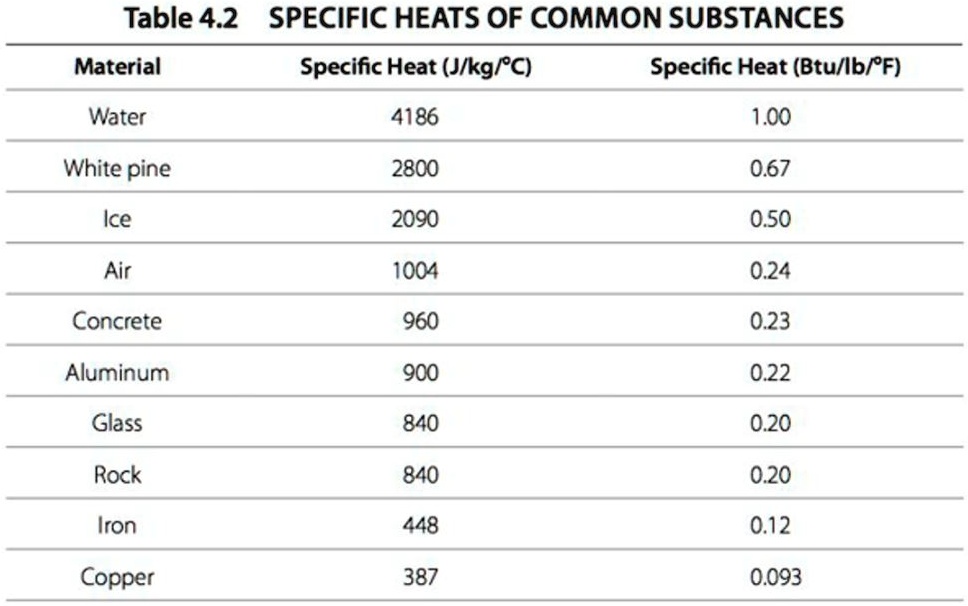
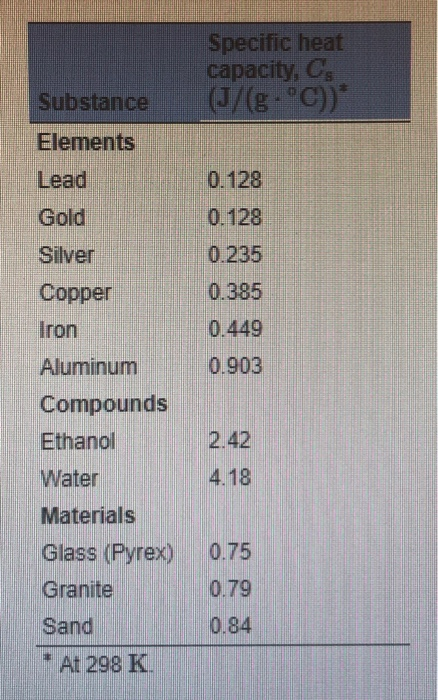
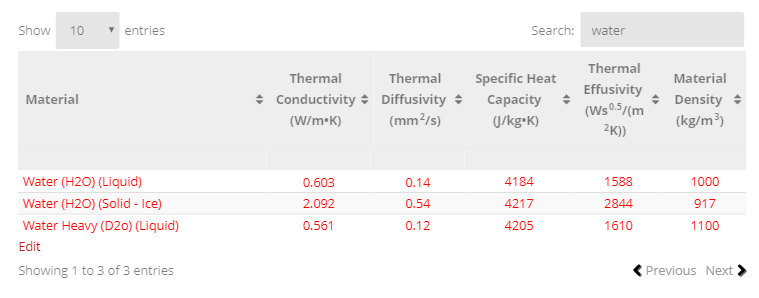
SOLVED: Table 4.2: SPECIFIC HEATS OF COMMON SUBSTANCES Material Specific Heat (J/kg°C) Specific Heat (Btu/lb°F) Water 4186 1.00 White pine 2800 0.67 Ice 2090 0.50 Air 1004 0.24 Concrete 960 0.23 Aluminum 900 0.22 Glass 840 0.20 Rock 840 0.20 Iron 448 ...