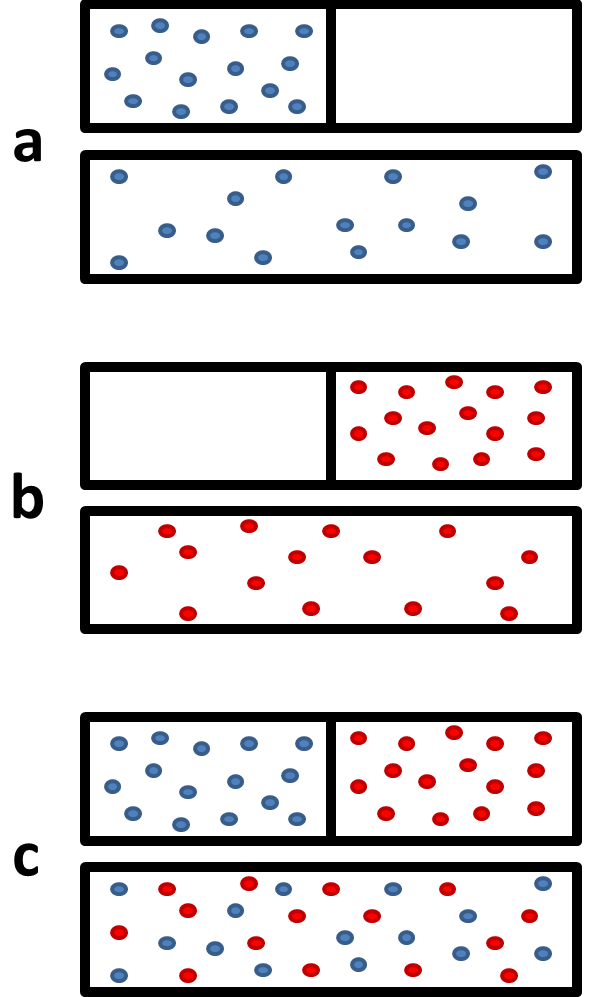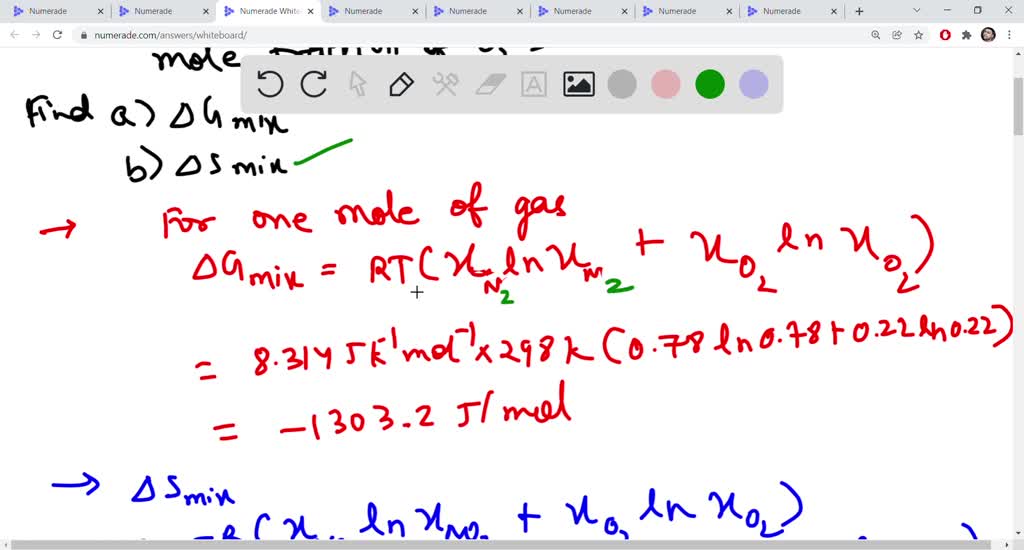
SOLVED: Calculate the (a) molar Gibbs energy of mixing, (b) the molar entropy of mixing when the two major components of air (Nz and 02) are mixed at 298 K The mole

Excess molar Gibbs energy of the binary mixture |Chemical Engineering Thermodynamics | GATE 2020 - YouTube

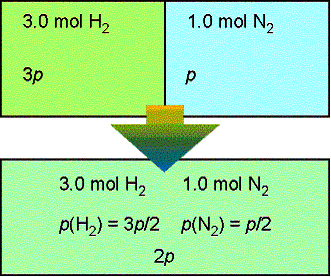
Now we are going to start looking at models for the chemical potential i of a given component i in a mixture The first model is the ideal gas mixture. -

SOLVED: 10.1 From Flory-Huggins theory the partial molar Gibbs free energy of 'mixing AG, with respect to the solvent for the formation of a polymer solution is given by AG, = 4-4" =