Vinylation of Aromatic Halides Using Inexpensive Organosilicon Reagents. Illustration of Design of Experiment Protocols | Journal of the American Chemical Society

Vinylation of phenol by acetaldehyde: A new reaction for the synthesis of o-vinylphenol - ScienceDirect

Enantioselective α-Vinylation of Aldehydes via the Synergistic Combination of Copper and Amine Catalysis | Journal of the American Chemical Society
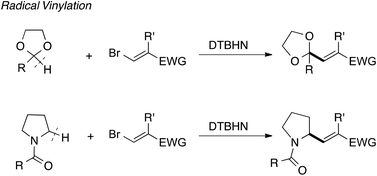
Radical vinylation of dioxolanes and N-acylpyrrolidines using vinyl bromides - Organic Chemistry Frontiers (RSC Publishing)

General Reaction Conditions for the Palladium-Catalyzed Vinylation of Aryl Chlorides with Potassium Alkenyltrifluoroborates
![PDF] Vinylation of a Secondary Amine Core with Calcium Carbide for Efficient Post-Modification and Access to Polymeric Materials | Semantic Scholar PDF] Vinylation of a Secondary Amine Core with Calcium Carbide for Efficient Post-Modification and Access to Polymeric Materials | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/f286ceb376de35cacaa09ad45f90c27e91883fe4/3-Table1-1.png)
PDF] Vinylation of a Secondary Amine Core with Calcium Carbide for Efficient Post-Modification and Access to Polymeric Materials | Semantic Scholar
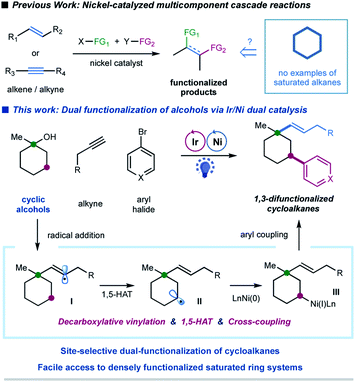
Sequential C–O decarboxylative vinylation/C–H arylation of cyclic oxalates via a nickel-catalyzed multicomponent radical cascade - Chemical Science (RSC Publishing) DOI:10.1039/D0SC01471K

Direct Vinylation of Alcohols or Aldehydes Employing Alkynes as Vinyl Donors: A Ruthenium Catalyzed C−C Bond-Forming Transfer Hydrogenation | Journal of the American Chemical Society

Plausible mechanism for the vinylation of alcohols and phenols in the... | Download Scientific Diagram

Enantioselective α-Vinylation of Aldehydes via the Synergistic Combination of Copper and Amine Catalysis

Low pressure vinylation of aryl and vinyl halides via Heck–Mizoroki reactions using ethylene - ScienceDirect
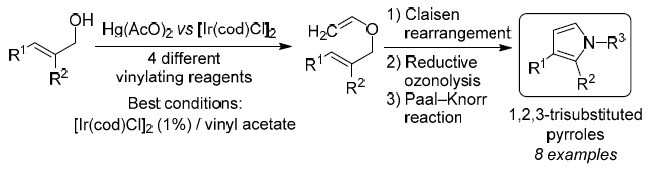
Synthesis of 1,3- and 1,2,3-functionalized pyrroles via Ir(I)-catalyzed vinylation of allyl alcohols | SpringerLink

Use of Iridium‐Catalyzed Transfer Vinylation for the Synthesis of Bio‐Based (bis)‐Vinyl Ethers - Spiegelberg - 2022 - Advanced Synthesis & Catalysis - Wiley Online Library

Vinylation of aromatic halides using inexpensive organosilicon reagents. Illustration of design of experiment protocols. - Abstract - Europe PMC
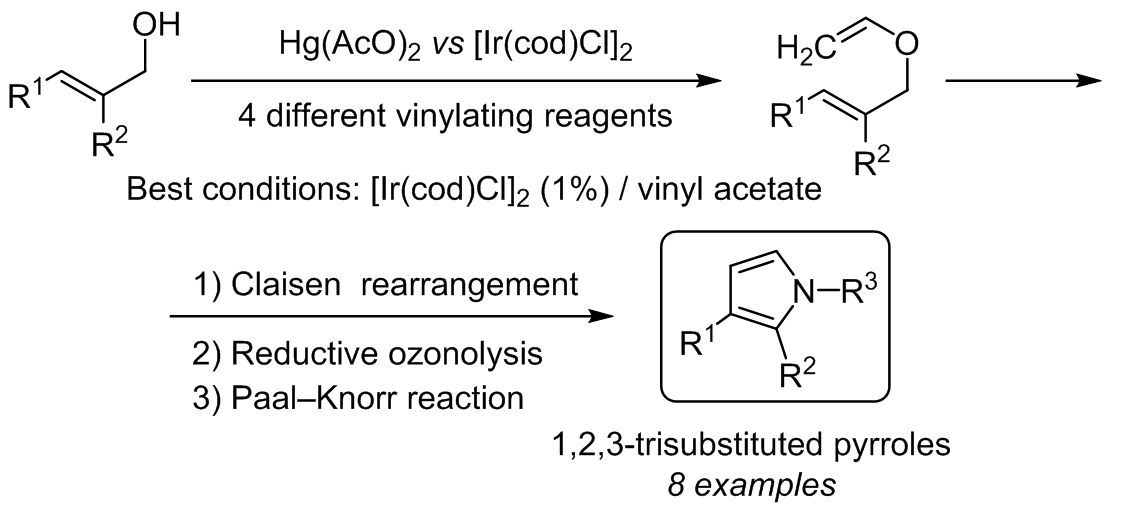
SYNTHESIS OF 1,3- AND 1,2,3-FUNCTIONALIZED PYRROLES <i>via</i> Ir(I)-CATALYZED VINYLATION OF ALLYL ALCOHOLS | Alavez-Rosas | Chemistry of Heterocyclic Compounds

![O‐Acylative Vinylation of Cyclodextrin‐Based [3]Rotaxane Towards Rotaxane Crosslinkers | GDCh.app O‐Acylative Vinylation of Cyclodextrin‐Based [3]Rotaxane Towards Rotaxane Crosslinkers | GDCh.app](https://chemistry-europe.onlinelibrary.wiley.com/cms/asset/08ecd971-9872-4012-a161-9b5d5db76568/ejoc202300171-toc-0001-m.png)