Teva and MedinCell Looking to Enter Increasingly Competitive LAI Schizophrenia Market - Clinical Trials Arena
Köp Risperidone Teva GmbH Pulver och vätska till injektionsvätska, depotsuspension 37,5 mg Risperidon 1 styck på Kronans Apotek | Kronans Apotek

FDA declines to approve Teva-MedinCell's risperidone injection for treating schizophrenia | Seeking Alpha

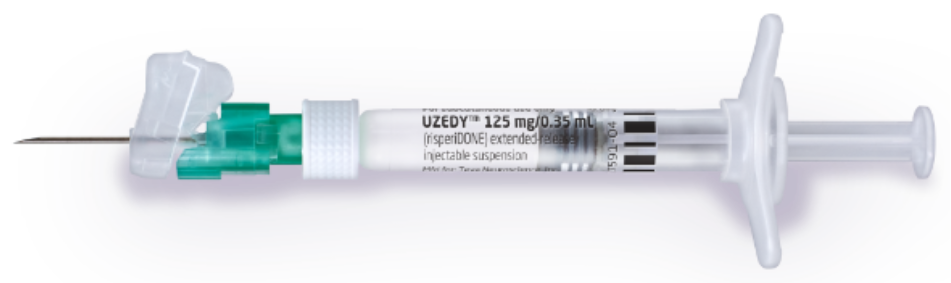
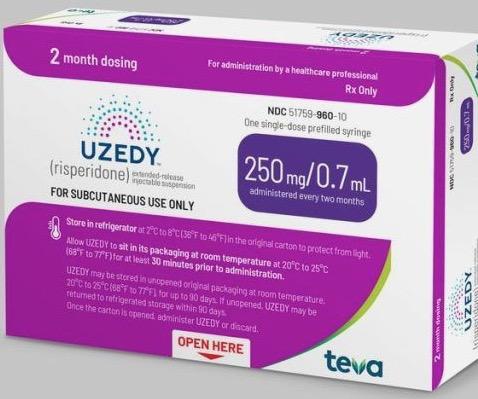
FDA Approves Uzedy (risperidone) Extended-Release Injectable Suspension for the Treatment of Schizophrenia in Adults

Risperidone "Teva GmbH" Teva GmbH pulver og solvens til depotinjektionsvæske, suspension 25 mg, 37,5 mg og 50 mg

Teva and MedinCell Announce FDA Approval of UZEDY™ (risperidone) Extended-Release Injectable Suspension, a Long-Acting Subcutaneous Atypical Antipsychotic Injection, for the Treatment of Schizophrenia in Adults

FDA Approves Uzedy (risperidone) Extended-Release Injectable Suspension for the Treatment of Schizophrenia in Adults-CliniExpert